How to Dispose of Medical Equipment the Right Way in 2026

When it's time to retire old medical and lab equipment, it's not as simple as just wheeling it out the door. Disposing of these assets is a high-stakes process where patient data, environmental regulations, and public safety all intersect. Get it wrong, and you could be looking at staggering fines and a damaged reputation, whether you're a local clinic or part of a nationwide healthcare system.
The right way to dispose of medical equipment demands a clear, documented process. This isn't just about a spring clean-out; it's about a systematic approach that covers inventory, risk assessment, proper decontamination, certified data destruction, and partnering with a vendor who knows the local and federal rules.
Your Roadmap for Compliant Medical Equipment Disposal
This guide is designed to cut through the noise and give you a clear roadmap for managing medical and lab equipment disposal from start to finish. For facilities in specific cities like Atlanta or across the country, we’ll share the strategies that separate responsible organizations from those taking a major risk.
The dangers of improper disposal are very real. An old diagnostic machine might still hold thousands of patient records, making it a goldmine for data thieves. We've seen centrifuges headed for the scrap heap that were never decontaminated, posing a serious biohazard risk to waste handlers and the public. These aren't just hypotheticals—they are liabilities that facilities in every state face every single day.
The Core Disposal Process: A Quick Overview
The entire journey can be broken down into three core stages: assessment, decontamination, and final disposition. Each step is designed to tackle specific risks, from data security breaches to environmental contamination.
This flowchart breaks down the essential steps for how to dispose of medical equipment the right way, creating a structured, risk-first approach.
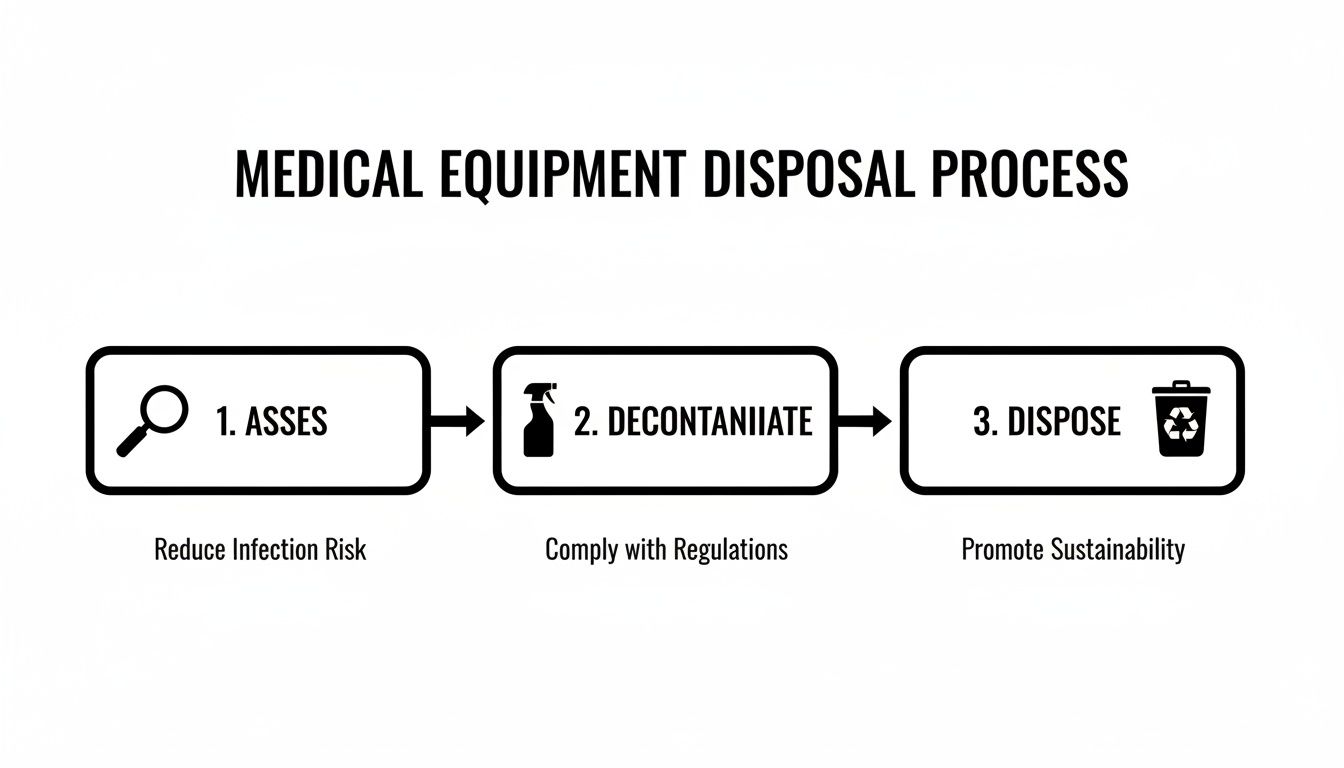
To give you a clearer picture, here is a summary of the main stages and their goals.
Medical Equipment Disposal At a Glance
| Stage | Key Action | Primary Goal |
|---|---|---|
| Assessment | Inventory equipment and identify all potential risks (data, biological, chemical). | Create a clear plan and understand the specific handling requirements for each asset. |
| Decontamination | Clean and sanitize equipment according to its use and regulatory standards. | Eliminate biological and chemical hazards to ensure safety for handlers and the public. |
| Disposition | Securely transport, destroy data, and recycle or dispose of the physical asset. | Ensure final disposal is compliant, documented, and environmentally responsible. |
This table maps out the fundamental workflow that keeps your facility safe and compliant, no matter your location.
The biggest mistake facilities make is treating all equipment the same. A retired office PC and a blood analyzer from a virology lab require completely different disposal protocols. A one-size-fits-all approach is a recipe for compliance failure.
To build a program that works, your team has to see these distinctions from the very beginning. This guide will give you the practical knowledge to do just that, helping you create a system that is both effective and defensible when it matters most.
Your First Step: Equipment Inventory and Risk Assessment

Before a single plug is pulled or any hardware gets moved, your project has to start with a detailed inventory. This isn’t just about creating a simple list; this is the bedrock of your risk management strategy, and it dictates every single thing that happens next.
Getting this part right is what separates a smooth, compliant disposal project from a chaotic and costly mess. This initial legwork is your first line of defense against compliance headaches and unexpected liabilities down the road.
Building Your Actionable Inventory Log
Fire up a spreadsheet or your asset management system—it’s time to track the essential data for every single item. Don't skip the details here. What seems like a minor point now can snowball into a massive problem later.
Your log needs to capture a full profile for each piece of equipment:
- Asset Type & Model: Is this a tabletop centrifuge, a massive MRI, or just an admin PC?
- Serial Number & Asset Tag: This is your unique identifier for tracking the asset from its current location to its final-disposition report.
- Current Condition: Be honest. Is it working, non-functional, or only good for spare parts?
- Location: Note the specific room, department, or lab. This is crucial for tracing potential contamination history.
- Data Storage: Does it have a hard drive, a memory card, or any other component that could hold sensitive information?
Once you have this catalog, you’re ready for the most important part of this phase: classifying by risk.
From Inventory to Risk-Based Classification
With your inventory complete, the next job is to assign a risk profile to each asset. This is where you figure out the exact handling requirements and categorize everything based on the potential hazards it might present.
A classic mistake we see all the time is treating all equipment as generic "e-waste." A centrifuge that’s been spinning samples in a bio-lab carries fundamentally different risks than a front-office computer, and your plan needs to reflect that reality from the very beginning.
By following a complete, step-by-step risk assessment process, you can systematically pinpoint and address these hazards before they ever become a real issue. This creates a clear, auditable trail that demonstrates due diligence.
A Real-World Scenario: University Lab Decommissioning
Let's walk through an example. A university is clearing out an older biology research lab. The inventory is a real mixed bag of complex instruments and basic hardware. Here's how they would break it down:
- High-Risk Biohazard: A mass spectrometer and several centrifuges were used for infectious disease research. These get tagged immediately for specialized, validated decontamination by a certified team before they can even be touched for removal.
- Moderate-Risk Chemical: Fume hoods and chemical storage cabinets have likely residual contamination. These require professional cleaning and a clearance certificate before they can be dismantled and sent off to be recycled as scrap metal.
- Data Security Risk: The lab’s workstations, servers, and even some diagnostic machines hold years of research data. These are flagged for DoD 5220.22-M 3-pass data wiping. Any drives that are non-functional or can't be wiped are scheduled for physical destruction.
- Low-Risk General E-Waste: Things like monitors, keyboards, and printers with zero data or bio-exposure are easy. They’re simply categorized for standard, responsible electronics recycling.
This risk-based approach ensures your resources go where they’re needed most. You avoid over-processing low-risk items while guaranteeing the high-risk assets get the specialized attention they demand. It transforms a potentially chaotic clean-out into a structured, compliant, and defensible project.
For a deeper dive into this part of the process, you might find our guide on https://www.scientificequipmentdisposal.com/lab-equipment-decommissioning-services-explained/ helpful. This categorization becomes the blueprint for your entire disposal strategy, ensuring every piece of equipment is handled safely and correctly from your facility to its final destination.
Mastering Decontamination and Waste Segregation

Before any equipment leaves your facility, proper decontamination is the absolute, non-negotiable first step. This is where we see facilities make their most costly—and dangerous—mistakes. Get this wrong, and you put your staff, your disposal vendor, and the public at serious risk.
The level of cleaning required boils down to the risk profile you identified earlier. An IV pole or administrative cart might just need a good wipe-down with an EPA-approved disinfectant. But for equipment coming out of a surgical suite or an infectious disease lab? That demands a much more serious approach.
Matching Decontamination to Equipment Risk
Think of decontamination as a spectrum. Not every piece needs the most intense sterilization, and knowing the difference saves you time and money. The goal is to match the method to the real-world risk.
- Low-Risk Items: Think waiting room furniture or office equipment. These items have no patient contact and typically just need a thorough cleaning to handle dust and grime. Your internal team can usually manage this.
- Medium-Risk Items: This includes devices with incidental patient contact, like blood pressure cuffs or EKG machines. They require cleaning followed by a proper disinfection step using hospital-grade chemicals.
- High-Risk Items: Anything used in invasive procedures or directly exposed to bodily fluids, pathogens, or other biohazards must be fully decontaminated. In many cases, this means full sterilization.
To get this right, you need to understand the detailed process of how to sterilize medical equipment to meet all safety standards and make these items safe for anyone to handle.
The Critical Importance of Waste Segregation
Once an item is clean, the next big challenge is segregation. This is where you can find huge cost savings. One of the most common errors we see is facilities incorrectly sorting their waste streams, which inflates disposal costs and multiplies compliance headaches.
Here’s the truth: a lot of what gets treated as hazardous waste isn't. Each surgical operation generates around 12 kilograms of waste, and operating rooms can account for up to 33% of all hazardous healthcare waste. But studies show that up to 90% of materials thrown into infectious waste bins could be safely handled as regular, non-hazardous waste. Misclassifying this carries a massive financial and environmental price tag.
Your team has to be trained to confidently tell the difference between these key categories:
- Regulated Medical Waste (RMW): This is anything saturated with blood or other potentially infectious materials (OPIM), along with all sharps and pathological waste. These items belong in designated red bags or sharps containers for specialized treatment, period.
- Chemical Waste: Solvents, reagents, and lab cleaning agents need to be segregated based on their specific hazards, like flammability or corrosivity. Never, ever mix different chemical waste streams together. Our guide on EPA-compliant laboratory equipment disposal offers more clarity on this.
- Electronic Waste (E-waste): This is the decontaminated equipment itself—monitors, circuit boards, and power supplies. These are destined for a certified electronics recycler, not a medical waste incinerator or a landfill.
- General Solid Waste: After decontamination and data sanitization, many components like metal casings or plastic shells can be disposed of as general refuse or scrap metal, depending on your local rules.
According to OSHA's Bloodborne Pathogens standard (29 CFR 1910.1030), employers must ensure that the worksite is maintained in a clean and sanitary condition. This includes implementing a written schedule for cleaning and an appropriate method of decontamination for all surfaces, tools, and equipment. A documented process is not just a best practice—it's a legal requirement nationwide.
Simply getting this sorting step right is one of the single most effective ways to slash disposal costs and run a more sustainable program. You stop paying for expensive regulated disposal on items that don't need it.
Ensuring Total Data Security and HIPAA Compliance
In healthcare and research, a data breach isn't just a headache—it's a full-blown crisis. When you're getting rid of old medical or lab equipment, protecting the patient data on those devices is your absolute top priority. It's not just good practice; it's a legal and ethical mandate under federal and state law.
One slip-up can lead to staggering HIPAA fines, a shattered reputation, and a permanent loss of patient trust.
The process of truly sanitizing data goes way beyond just hitting a "delete" button. You need a certified, documented process that proves every last bit of electronic Protected Health Information (ePHI) has been completely and permanently erased. This is where so many facilities, even with the best intentions, find themselves in hot water.

Making Sense of NIST 800-88 Data Wiping Standards
The gold standard for this process comes from the National Institute of Standards and Technology (NIST). Their Special Publication 800-88 is the go-to guide for media sanitization, and it lays out a few different ways to get the job done.
Choosing the right method really comes down to how sensitive your data is and what kind of device you're dealing with. It's critical to understand the difference.
To help you decide, here’s a quick breakdown of the most common data sanitization methods and where they fit into a compliant disposal plan.
Data Sanitization Methods Explained
| Method | Description | Best For | Compliance Level (HIPAA/NIST) |
|---|---|---|---|
| Clear | Uses software to overwrite data once. It’s like erasing a whiteboard. | Low-risk, non-sensitive data on functional devices. | Not sufficient for ePHI. Easily recoverable. |
| Purge | Overwrites data multiple times with complex patterns (DoD 5220.22-M 3-pass, for example). Data recovery is infeasible even with advanced lab tools. | Most devices containing ePHI, financial data, or other sensitive information. | Meets HIPAA & NIST requirements for most media. |
| Destroy | The media is physically obliterated through shredding, pulverizing, or incineration. | Damaged hard drives, SSDs, mobile devices, and any media that can’t be reliably purged. | The highest level of security. Exceeds all compliance requirements. |
Understanding these methods is the first step, but putting them into practice correctly is what truly matters for compliance.
It's important to know that HIPAA doesn't tell you exactly which disposal method to use. The Security Rule just requires that you have policies and procedures to "render ePHI unreadable, indecipherable, and otherwise cannot be reconstructed." Following the NIST guidelines is the universally accepted way to prove you've done just that.
Don't Overlook Hidden Data Liabilities
One of the biggest mistakes we see is facilities focusing only on computers and servers. The truth is, modern medical equipment is packed with hard drives, memory cards, and onboard storage that can hold a treasure trove of patient data.
Your IT and compliance teams need to be detectives, hunting down these hidden data risks. Think about it—every piece of equipment with a screen, a memory card slot, or a network port is a potential data breach waiting to happen.
Use this checklist as a starting point for your inventory:
- Diagnostic Imaging Systems: MRIs, CT scanners, and X-ray machines often have massive internal hard drives storing thousands of patient scans.
- Lab Analyzers: Blood analyzers, mass spectrometers, and genetic sequencers can hold test results tied directly to patient names and records.
- Patient Monitoring Systems: Those bedside monitors and telemetry units? They frequently cache patient vitals and other real-time sensitive data.
- Infusion Pumps: So-called "smart pumps" can retain detailed logs of medication administration, including patient-specific dosing information.
If a device isn't properly sanitized, you're leaving the door wide open for a compliance nightmare. For a more detailed look at how these methods apply to your specific equipment, you can learn more about our certified secure data destruction services.
Finally, your job isn't done until you have the paperwork to prove it. Always, always demand a Certificate of Data Destruction from your disposal vendor. This document is your official record that every piece of data-bearing media was sanitized according to industry standards—an essential piece of evidence for any compliance audit.
How to Choose a Compliant Disposal Partner
When that old medical equipment leaves your loading dock, your liability doesn't leave with it. In many ways, it's just getting started. Picking the right disposal vendor is probably the single most important decision you'll make in this process. This is the point where all your careful planning is either validated or completely worthless.
A cheap scrap hauler might give you a tempting price, but the hidden costs from a data breach, an environmental fine, or a compliance failure can be staggering. A truly compliant partner is different. They act like an extension of your own risk management team, making sure the job gets done right and giving you the paper trail to prove it.
Start with the Non-Negotiable Certifications
The very first filter for any potential partner should be their certifications. These aren't just nice-to-have badges they can put on their website; they are hard-earned proof of a commitment to security, safety, and environmental standards, all verified by third-party auditors.
If a vendor can’t show you at least one of these two certifications, you should walk away. It's that simple.
- R2 (Responsible Recycling): The R2 Standard is the gold standard for electronics recyclers. It covers everything from protecting the environment and ensuring data is destroyed to the health and safety of their own workers. An R2-certified facility guarantees they aren't just going to illegally ship hazardous e-waste overseas.
- e-Stewards: This is another top-tier global certification for electronics recyclers. It's known for being incredibly strict, with a complete ban on exporting hazardous e-waste to developing nations or dumping it in landfills.
Having these certifications is your assurance that the vendor has a proven, audited system for handling the complexities of medical and lab equipment disposal.
Critical Questions for Your Potential Partner
Once you've confirmed a vendor has the right certifications, it's time to dig a little deeper. Any professional outfit will welcome these kinds of questions and should have clear answers ready. This is how you separate the real experts from the amateurs.
Insurance and Liability
A vendor's insurance is what protects you if something goes wrong during transport or processing.
- Do you carry pollution liability insurance? What are the coverage limits?
- Can you provide a certificate of insurance that lists our organization as an additional insured?
- What are your general liability and workers' compensation coverage levels?
Chain of Custody and Documentation
This paperwork is your proof of compliance.
- Can you show me a sample of your chain-of-custody documents?
- Do you provide a Certificate of Recycling and a Certificate of Data Destruction for every single job?
- How do you track our assets from the moment you pick them up to their final disposition?
Downstream Vendor Network
You have to know where your equipment actually ends up.
- Who are your downstream recycling partners? Are they also R2 or e-Stewards certified?
- Can you provide a full downstream audit trail if we request it?
A reliable partner views your equipment as a chain of responsibility, not just a commodity. They understand that their actions directly reflect on your organization’s reputation and compliance standing.
This global sense of responsibility is more important than ever. The challenges with medical equipment donations in some parts of the world show exactly why professional logistics are vital. For instance, about 80% of medical devices in sub-Saharan Africa are donated. Yet, the World Health Organization finds that up to 70% of them are not used effectively, often due to a lack of parts, training, or proper disposal channels. A key solution being explored is "reverse logistics," where donors commit to taking back expired devices. You can explore the full findings on responsible device lifecycle management to see the scale of the problem.
Scenario: The Tale of Two Vendors
Let's imagine a hospital in the Atlanta metro area needs to get rid of a dozen old patient monitors.
- Vendor A (The Scrap Hauler): They offer to pick up the monitors for free. They have no certifications, provide zero paperwork, and just say they'll "take care of it." Three months later, an investigator finds one of the monitors in a local landfill with the hospital's asset tag still attached. The hospital is now facing fines and a PR nightmare.
- Vendor B (The Certified Partner): They provide a clear quote that covers secure transport, certified data destruction, and documented recycling. They show proof of their R2 certification and pollution liability insurance right upfront. A week after pickup, the hospital receives a Certificate of Data Destruction and a Certificate of Recycling, officially closing the loop and ending their liability.
The right choice is obvious. For organizations in Atlanta and across the country, a partner with real logistical expertise and a deep understanding of state-specific environmental laws is an asset. Whether it's a full lab cleanout or just a routine pickup, choosing a certified e-waste recycling company is the final, crucial step to protect your organization.
The Business Case for Responsible Equipment Disposal
Thinking about how to dispose of medical equipment the right way isn't just about ticking a compliance box. It's a strategic move that directly protects your budget and your brand. When you shift your perspective, you stop seeing disposal as a pure cost and start seeing it for what it is: a critical investment in risk management and corporate responsibility.
This approach delivers real, tangible financial returns. For instance, we often see facilities overspending because they don't get waste segregation right. Paying for expensive regulated medical waste handling for an item that only needed standard e-waste recycling is an incredibly common—and costly—mistake. Mastering asset classification can slash your disposal costs.
Boosting Your ESG Profile
Beyond the direct savings, responsible disposal is a powerful way to build your organization's reputation. These days, there’s enormous pressure on hospitals, labs, and universities to improve their Environmental, Social, and Governance (ESG) scores. Everyone from investors and patients to the local community is paying close attention to these metrics.
This is where partnering with a certified recycling vendor gives you a clear win. A compliant partner can provide the detailed sustainability reports that quantify your positive impact, including:
- Total weight of materials recycled and successfully diverted from landfills.
- Breakdowns of recovered commodities like steel, aluminum, and plastics.
- Certified proof of secure data destruction, which strengthens your governance credentials.
These reports arm you with the hard data you need to back up your ESG claims and show a real commitment to doing the right thing.
Thinking about disposal solely in terms of cost misses the bigger picture. A well-managed program is an investment that pays dividends in risk mitigation, brand enhancement, and stakeholder trust, whether you're a local clinic in Atlanta or a national healthcare system.
A Growing Market Reflects a Growing Responsibility
The demand for compliant disposal solutions isn't just a passing trend; it's a market that's expanding at a breakneck pace. As of 2025, the global medical waste management market was valued at around USD 16.7 billion, and forecasts show it rocketing to USD 34.06 billion by 2030. This explosive growth is driven by the sheer volume of medical waste being generated and a much-needed rise in awareness around compliant practices.
This market boom highlights a crucial point: facilities nationwide are actively looking for better solutions. When you adopt a proactive and responsible disposal strategy, you're not just keeping up—you're positioning your organization as a leader. You can find more information about what this involves by checking out our guide on how to establish a medical equipment recycling program.
Ultimately, a smart disposal plan protects your budget, safeguards your reputation, and proves you’re committed to ethical operations—a message that resonates with everyone you serve.
Your Medical Equipment Disposal Questions, Answered
Even the most buttoned-up process leaves a few questions. We get a lot of the same ones from facility managers and compliance officers nationwide, so we’ve put together some straight answers.
Here’s what people ask us most about getting rid of medical equipment the right way.
What Paperwork Should I Keep After Disposal?
This is non-negotiable. For any potential audit, you need a solid paper trail.
Make sure your vendor provides, and you securely file:
- A complete chain-of-custody document.
- A Certificate of Disposal or Recycling.
- A Certificate of Data Destruction for any device that stored information.
These documents are your legal proof that you followed every compliance step to protect patient data and the environment.
Can We Donate Old Medical Equipment That Still Works?
Donating is a great idea, but it’s not as simple as dropping it off. You have to do your homework. The equipment must be fully decontaminated, all data needs to be professionally wiped clean according to NIST standards, and you have to confirm it's both functional and safe.
A word of caution: Don’t just give it away. Work with reputable charities that can actually verify the end user can use and maintain the equipment. Dumping broken or unserviceable gear on a well-meaning nonprofit can become a huge liability for you and a burden for them.
What's the Biggest Mistake You See Facilities Make?
Hands down, the most common and costly mistake is picking a disposal vendor based on price alone. We see it all the time with facilities across the U.S. A cheap quote is tempting, but it almost always means they’re cutting corners somewhere.
Usually, this means they lack key certifications like R2 or e-Stewards. This leads to sloppy data handling, environmental violations, and a complete lack of auditable paperwork. The fallout can be severe, exposing your organization to massive legal, financial, and reputational hits. Always, always put compliance and certification before a rock-bottom price.
Does Equipment Have to Be Working to Be Recycled?
Not at all. A certified e-waste recycler is set up specifically to handle non-functional and obsolete medical equipment. Their job is to dismantle these items, recover valuable materials like metals and plastics, and safely dispose of any hazardous components.
The key is to use a certified recycler, not a generic scrap hauler. That's the only way to guarantee your old equipment doesn't just end up in a landfill, which is the exact opposite of proper medical equipment disposal.
Navigating the complexities of equipment disposal requires a partner who understands the stakes. Scientific Equipment Disposal provides certified, compliant, and documented solutions for hospitals, labs, and universities throughout the Atlanta metro area and nationwide. We handle everything from on-site pickup to certified data destruction, ensuring your assets are managed responsibly from start to finish. To get a clear, straightforward plan for your equipment, visit us online for a disposal assessment.