Scientific Equipment Recycling Programs: A How-To Guide

A lot of recycling programs start the same way. A lab closes, a renovation date gets moved up, a PI wants old instruments gone, or IT flags aging devices with stored data. Then facilities gets pulled in, EHS asks for decontamination status, procurement wants to know whether anything still has value, and nobody wants to be the person who approved the wrong disposal path.
That’s where Scientific Equipment Recycling Programs either become disciplined asset recovery projects or expensive cleanup jobs with compliance risk attached. In Atlanta, I see the same pressure points across hospitals, universities, research groups, and corporate labs. The environmental case matters, but it usually isn’t the issue holding up approval. The key questions are simpler. Who owns the decision, what’s the financial outcome, and how do you prove the liability left your organization the right way?
Labs generate waste at a scale most general office recycling plans were never designed to handle. According to My Green Lab’s waste overview, laboratories worldwide generate up to 5.5 million metric tons of plastic waste annually, nearly 2% of global plastic waste production, and research facilities can produce up to 12 times more waste per square foot than typical office spaces. That’s the backdrop. The practical work is turning a pile of mixed lab assets into a documented, secure, financially defensible disposition program.
Establishing Program Goals and Assembling Your Team
The first mistake is starting with a pickup date.
The right starting point is deciding what success means for your facility. In one building, the top priority is data security because analyzers, lab PCs, and attached storage may contain regulated information. In another, the priority is clearing a floor fast enough to stay on construction schedule. In a university setting, the biggest win may be reuse, donation, or resale before recycling even starts.

Set priorities in the right order
A workable program usually balances four targets:
- Security first: Any asset with storage media, network connectivity, or patient-related workflow history needs to be identified before it leaves the room.
- Compliance second: Equipment exposed to chemicals, biological materials, or controlled environments needs the right decontamination paperwork and internal release approvals.
- Financial recovery third: Some assets should be remarketed, some donated, and some dismantled for materials. Mixing those categories too early causes lost value.
- Operational speed fourth: Loading docks, elevator access, shutdown sequencing, and building rules affect cost more than is commonly expected.
If your organization hasn’t documented these priorities, your recycler ends up making decisions your internal team should have made.
Practical rule: If legal, EHS, and IT haven’t agreed on release conditions before removal starts, the project is still in planning, even if the trucks are scheduled.
Build a team that can actually approve decisions
Scientific equipment recycling programs fail when they’re managed by only one department. Facilities might control access and timing, but they usually can’t sign off on media sanitization. IT can clear drives, but they don’t decide whether a biosafety cabinet is ready for transport. Procurement may know asset history, but not contamination status.
Use a small working group with clear authority:
- Lab manager or department lead handles equipment identification, use history, and owner confirmation.
- IT or information security flags data-bearing assets and sets sanitization requirements.
- EHS reviews contamination risk, decontamination records, and release conditions.
- Facilities or project management controls site access, dock scheduling, and shutdown sequencing.
- Finance or procurement checks ownership, lease status, and any resale or write-off implications.
For larger decommissions, assign one person to control the master asset list. Not five versions of a spreadsheet. One list.
Match the program to the site
A hospital lab in metro Atlanta doesn’t need the same operating plan as a university chemistry department. Hospital teams usually need tighter chain-of-custody expectations and faster signoff on data-bearing devices. Universities often have a wider mix of surplus conditions, from reusable teaching equipment to obsolete instruments that only have scrap value.
That’s why early scoping matters. A broad intake review of laboratory equipment categories commonly handled in decommission projects helps teams separate routine removals from assets that need special handling.
A good kickoff meeting ends with three things written down: which assets are in scope, who can release them, and what outcome each category should follow. Without that, the rest of the process turns into reactive problem solving.
Navigating the Critical Compliance and Regulatory Checklist
A lab shutdown can stay on schedule for weeks, then stall over one missing release form, one analyzer with an old hard drive, or one freezer that no one can certify as decontaminated. That is usually how compliance problems start in Atlanta-area decommissions. Not with intentional misconduct, but with loose documentation and assumptions about who was handling what.
That risk increases for health systems, universities, and commercial labs operating across multiple sites. As noted by Beyond Surplus on laboratory equipment disposal compliance gaps, organizations run into trouble with liability transfer, inconsistent state documentation requirements, and weak visibility into downstream processing. For lab directors and facility managers, the business issue is clear. If the records are weak, liability stays close to the generator, and recovery value gets harder to defend internally.
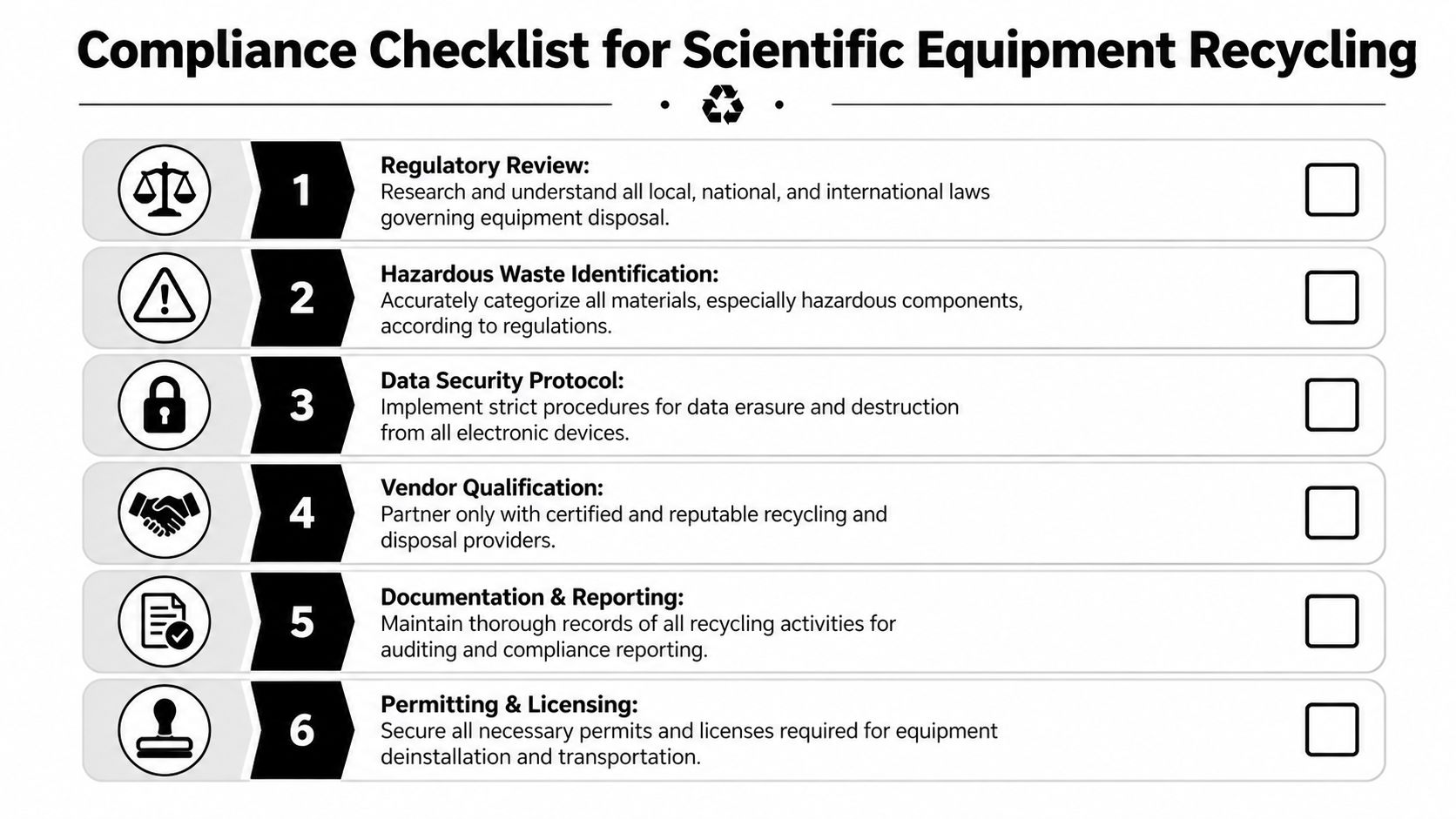
Define data handling before pickup
Data risk has to be addressed before trucks are scheduled. I have seen removals delayed because an instrument looked like scrap, then turned out to contain a control PC, embedded storage, or removable media tied to research, patient, or calibration records.
Scientific equipment with hidden data risk often includes analyzers, imaging systems, chromatography platforms, attached workstations, instrument servers, and older standalone PCs that remained in the room after software support ended.
For data-bearing assets, set the rules in writing:
Identify the storage type
Confirm whether the asset contains a hard drive, solid-state media, removable media, or embedded memory.Assign the sanitization path
Specify whether the media will be software-sanitized or physically destroyed, and define which standard your organization accepts.Link equipment and media records
Tie the host asset serial number to the media identifier so the final paperwork matches what left the site.Require asset-level proof
Certificates only help if they reference the specific assets or media removed from your facility.
This matters for compliance, but it also affects cost recovery. Equipment cleared for legal resale or parts recovery usually returns more value than equipment placed on hold because nobody could approve the data disposition.
Exposure history determines whether the asset can move
Two identical instruments can follow completely different disposition paths based on prior use. A centrifuge from a teaching lab is not handled the same way as one used in pathology, molecular diagnostics, or solvent-heavy research.
Before any item is wrapped, palletized, or removed, confirm four points:
- Known exposure history for chemicals, solvents, biohazards, radiological materials, or controlled substances
- Decontamination status and whether your internal policy requires a signed certificate
- Residual risk components such as tubing, traps, reservoirs, pumps, filters, or sample contact parts
- Authorized release from the department or compliance owner with authority to approve disposition
Schedules often slip. A crew arrives, the loading dock is booked, everyone is ready to clear the room, and then one unit has no documented use history. That unit stays. Sometimes the whole pickup does.
Equipment is releasable only when the exposure history and release conditions are documented.
Chain of custody protects both compliance and financial reporting
Chain of custody is not just an environmental or safety record. It supports audit defense, internal write-offs, insurance questions, and resale accounting. If your finance team wants to know why one group of assets generated recovery revenue and another group went to destruction, the answer has to be in the file.
At a minimum, require:
- Asset-level pickup details such as manufacturer, model, serial number, condition, and disposition category
- Transfer records with dates, signatures, and site of origin
- Transport traceability identifying the carrier or controlled fleet
- Downstream documentation showing who processed the material and under what disposition path
- Final disposition records covering destruction, recycling, resale, or parts recovery
A vendor that offers EPA-compliant laboratory equipment disposal processes should be able to explain exactly when custody changes, when liability changes, and what paperwork your team receives at each step. If they cannot explain that clearly, expect gaps later.
For teams also comparing removal contractors, the issue is similar to finding commercial moving companies. The lowest pickup price does not tell you much about documentation discipline, controlled handling, or downstream accountability.
Vendor qualification belongs on the compliance checklist
Permits matter. So does the vendor interview.
Ask direct questions and expect direct answers:
| Compliance area | Ask this question | What a strong answer sounds like |
|---|---|---|
| Data handling | How do you process failed drives and embedded storage | The vendor separates software sanitization from physical destruction and documents each path |
| Hazard release | What documentation do you require before pickup | The vendor has written release requirements by equipment type or exposure class |
| Downstream processing | Who receives material after pickup | The vendor can name qualified downstream processors and explain their role |
| Records | What final documents will we receive | The vendor specifies asset logs, transfer records, destruction documentation, and resale reporting where applicable |
| Multi-state work | How do you handle different jurisdiction requirements | The vendor uses a documented workflow instead of relying on verbal assurances |
The practical trade-off is simple. Tight controls take more time up front, but they reduce schedule disruption, rejected pickups, and audit exposure later. They also make ROI calculations more credible because your resale, recycling, and destruction categories are supported by records instead of estimates.
Executing Asset Inventory Deinstallation and Logistics
A lab shutdown usually starts the same way. One group wants the space cleared by Friday, another still needs access to two analyzers, facilities has a freight elevator window, IT is asking where the embedded drives are, and nobody fully agrees on which assets still have resale value. That is why deinstallation and logistics determine both project cost and liability exposure.

Start with an asset map, not a pile count
A useful inventory has to support removal, valuation, and final disposition. If the sheet only says “microscope” or “freezer,” the crew cannot plan handling correctly, and finance cannot separate resale candidates from scrap.
Build the inventory around details that change cost and risk:
- Condition: working, repairable, incomplete, obsolete, damaged
- Disposition path: reuse, resale review, parts recovery, recycling, destruction
- Data status: none known, contains media, sanitization required, destroy media
- Release status: cleared, pending decontamination, lease review, hold
- Physical handling needs: bench item, palletized unit, oversized, fragile, requires rigging support
This is also where ROI starts to become real. High value bench-top equipment, specialty detectors, and serviceable peripherals often lose recovery value when they get mixed into bulk e-waste gaylords. Keep those items identified at the room level, with accessories and power supplies matched to the parent asset.
Deinstallation has to protect the asset, the building, and the schedule
Good crews do more than disconnect equipment. They document what was removed, keep assemblies together, separate media-bearing devices, protect sensitive components, and avoid damage to door frames, casework, and freight paths.
Sequencing matters. In active labs, the cheapest plan on paper often becomes the most expensive plan in practice if removal starts before users finish final runs or before room release approvals are in hand.
A workable removal sequence usually looks like this:
Pull surplus and boxed equipment first
Clear storage rooms, retired peripherals, and obvious dead inventory to create staging space.Schedule shutdowns for active instruments
Confirm final use, data capture, and release timing with lab owners before disconnecting anything.Handle specialty assets as separate work orders
Large analyzers, incubators, cold storage units, and utility-connected systems need their own labor plan, packing method, and route review.Hold time for exceptions
Missing cords, software locks, unclear ownership, and incomplete decontamination records show up on nearly every project.
I have seen well-funded decommissions stall over simple misses. No pallet jack on the right floor. No approved dock window. No answer on whether a controller contains storage. Those details delay trucks, increase labor hours, and turn a recoverable asset into damaged scrap.
Logistics drives the real project budget
Transportation costs are rarely just transportation costs. Atlanta-area sites often require certificate of insurance review, dock scheduling, liftgate planning, after-hours access, and elevator reservations. Research buildings add another layer because access windows may have to avoid animal care, clinical activity, or shared core operations.
That is why lab removals need a tighter process than ordinary office relocations. Teams comparing vendors sometimes start by finding commercial moving companies to understand scheduling and access planning. That is useful background. Scientific equipment moves still require asset-level chain of custody, separation of data-bearing items, and documented handling for potentially exposed equipment.
Use a site logistics checklist before the first pickup date is confirmed:
- Dock and freight elevator reservations
- Approved building access times
- On-site staging area
- Packaging for fragile electronics, optics, and glass
- Pallets or crates for heavy equipment
- Separate containers for data-bearing assets
- Clear routing for items that require rigging or extra labor
- Pickup timing that avoids disruption to research or clinical work
That up-front planning has a direct financial effect. Fewer return trips, less waiting time, less damage, and better asset segregation usually improve net recovery and reduce avoidable service charges.
Controlled pickup sets up certified processing later
Downstream results depend on how material leaves the building. If the pickup crew loads mixed assets with weak labeling and no custody controls, the processor receives a bulk stream instead of a traceable inventory. At that point, resale value drops, reporting gets thinner, and your audit file gets harder to defend.
As noted earlier, qualified lab recycling programs rely on on-site review, clear categorization, controlled packing, and documented transfer into certified processing channels. In practical terms, the pickup team should know which items are for resale review, which require destruction, which need decontamination verification, and which must stay segregated because they contain storage media or regulated components.
For university cleanouts and multi-room shutdowns, specialized scientific equipment removal for labs and universities is materially different from generic hauling. The point is not just to get equipment onto a truck. The point is to preserve asset identity, protect recovery value where it exists, and maintain records that hold up if compliance or finance asks questions later.
Ensuring Secure Data Sanitization and Certified Disposition
Not all recycling is equal. Some vendors pick up equipment, issue a generic receipt, and disappear the details into a downstream network you never see. That may remove clutter from your building, but it doesn’t give your compliance team much to stand on if questions come later.
The need for qualified processing is only growing. According to Cognitive Market Research’s recycling equipment market report, the global market for recycling equipment is projected to reach $30.985 billion by 2025, yet only 17.4% of the 53.6 million metric tons of global e-waste generated in 2019 was properly collected and recycled. Bigger market, same core problem. Too much material still moves through channels that aren’t transparent enough.
Choose the sanitization method that fits the media
For data-bearing scientific and medical equipment, the disposition process begins with the storage media, not the metal housing.
Three methods come up most often:
- Software wiping works when the drive is functional and the device can be accessed reliably. For organizations that require documented overwrite procedures, this is often the preferred route when remarketing is still possible.
- Degaussing can be appropriate for certain magnetic media, but it also makes the drive unusable afterward.
- Physical shredding is the fallback when the drive is failed, obsolete, inaccessible, or not worth preserving.
A common mistake is treating all data destruction as interchangeable. It isn’t. If the asset might be reused, wiping may preserve value. If the media is compromised or the compliance requirement is stricter, shredding is cleaner and easier to defend.
Certification matters because downstream behavior matters
You don’t need a vendor that says the right words. You need one that can show where equipment goes after pickup and what standards govern that path.
That’s why certified disposition matters. When processors follow established standards and document material flows, your organization has a stronger basis for claiming that assets were managed responsibly. When they don’t, you may still get a pickup receipt, but you won’t have much visibility into export controls, component handling, or landfill avoidance.
If a recycler can’t explain the downstream chain in plain language, assume they can’t defend it under scrutiny either.
The same logic shows up outside lab settings. Consumer-focused guidance like selling your old laptop in Singapore reminds people to think about device condition, residual value, and secure data removal before disposal. The stakes are much higher in clinical and research environments, but the principle is the same. Value recovery only works if the data risk is closed first.
Final records are part of the service
A secure disposition process should end with documentation your legal, IT, and facilities teams can all use. That usually means asset-level logs and some form of final certificate for destroyed media or equipment categories requiring documented destruction.
A sample certificate of destruction format for scientific equipment and media disposition gives facilities managers a useful benchmark for what to ask for. The document should tie back to the assets removed, not just the project name.
Certified disposition also helps separate reuse from recycling. That distinction matters. Reuse can preserve value. Recycling can recover materials. Destruction closes the loop on nonfunctional or sensitive assets. Trouble starts when a vendor uses one word to describe all three.
Evaluating Program Success and Choosing Your Vendor
Facilities managers usually get asked two questions after a project is over. Did we stay compliant, and what did it cost us? The frustrating part is that many recycling programs still don’t provide a transparent way to answer the second question.
That gap is real. The University of Pennsylvania resource on lab supplies reuse and donation highlights a broader issue in the market: recycling content often lacks transparent ROI models, including total cost, hidden fees, and equipment valuation methods that facilities teams need when comparing recovery versus disposal. That’s why good vendor selection starts with financial clarity, not branding.
What success actually looks like
A solid scientific equipment recycling program should produce outcomes in four categories:
- Direct cost control through predictable pickup, packing, transport, and processing charges
- Cost avoidance by reducing exposure to bad data handling, undocumented disposal, and failed handoffs
- Asset recovery through reuse, resale, or parts harvesting where viable
- Administrative efficiency through complete records that close projects quickly
You don’t need a complicated finance model to compare vendors. You need line-item visibility.
Ask for separation between:
- onsite labor
- packing materials
- transportation
- data destruction services
- downstream processing
- revenue sharing, if any
- charges for nonstandard items or project delays
If a quote bundles everything into one vague project fee, it becomes hard to tell whether the economics are strong or just obscured.
Use a simple internal ROI worksheet
Most organizations can evaluate a program using a practical worksheet with three columns:
| Cost or value area | Questions to ask internally | What to document |
|---|---|---|
| Removal cost | What did labor, transport, and processing cost | Approved quote, change orders, final invoice |
| Avoided internal burden | What staff time, storage burden, or risk exposure did this remove | Internal labor notes, timeline impact, release records |
| Recovered value | Which assets were reused, sold, or diverted into a better channel than disposal | Asset list, value allocation method, final disposition records |
That won’t produce a perfect model, but it will produce a defensible one. If your organization needs a framework for comparing liquidation paths on mixed assets, DIYAuctions' guide to asset liquidation is a useful outside reference for thinking through value recovery versus straight disposal, especially when surplus equipment includes more than one asset class.
Vendor Selection Criteria Checklist
The best vendor interviews are structured. Don’t ask whether a provider is “full service.” Ask how the service works when something goes wrong.
| Criterion | What to Look For | Why It Matters |
|---|---|---|
| Data sanitization process | Clear explanation of wiping versus shredding and how each is documented | Protects regulated data and supports audit defense |
| Chain of custody | Asset-level tracking from pickup to final disposition | Shows where liability transfers and what happened next |
| Decontamination policy | Written requirements for equipment release and hazardous exposure documentation | Prevents rejected pickups and unsafe removals |
| Logistics capability | Ability to deinstall, pack, palletize, and remove from complex lab environments | Reduces project disruption and surprise costs |
| Downstream transparency | Named processing standards and identifiable downstream partners | Lowers risk of improper handling after pickup |
| Reporting quality | Detailed manifests, destruction records, and final disposition summaries | Helps finance, EHS, and IT close the project cleanly |
| Insurance and contractual clarity | Written terms on responsibility during handling and transport | Clarifies risk allocation during handoff |
| Multi-site consistency | Standard process across different facilities and states | Matters for hospital systems and university networks |
A vendor should be able to explain its process without relying on marketing language. If the answer sounds polished but not specific, keep asking.
For teams comparing providers, a practical benchmark is whether the company can show a repeatable workflow similar to what you’d expect from certified scientific equipment disposal providers. Certifications matter, but so do operational details. The strongest vendors can explain both.
Frequently Asked Questions About Equipment Recycling
What about small lab plastics and glassware
Handle these separately from electronic equipment loads. Lab plastics, tip boxes, conical tubes, and glassware usually follow a different waste stream than instruments and IT hardware. If there’s any contamination concern, route the decision through EHS before combining anything for pickup. Don’t assume “recyclable material” means it belongs in the same load as electronics.
How should leased equipment be handled
Don’t release leased equipment into a recycling program until ownership is confirmed. Review the lease, service agreement, or manufacturer return terms first. In many cases, the lessor controls end-of-term disposition, and removing the asset without approval creates an avoidable contract problem.
Can lab equipment and office IT go in the same project
Yes, but not as an undifferentiated pile. Mixed loads are common during consolidations and closures, especially in hospitals and universities. The fix is categorization. Keep standard office IT, scientific instruments, and anything with possible contamination history separated in the inventory and on the truck paperwork.
What if the equipment has no resale value
That’s normal. A lot of scientific equipment recycling programs involve obsolete, incomplete, damaged, or highly specialized assets that won’t attract a secondary buyer. The absence of resale value doesn’t reduce the need for secure handling, documented transport, and compliant final disposition.
Who should sign off before equipment leaves the building
At minimum, the asset owner or department lead, IT for data-bearing devices, and EHS when exposure history is relevant. Facilities often controls site logistics, but facilities alone usually shouldn’t be the only release authority for specialized lab equipment.
Do we need decontamination paperwork for every item
Not always. But if a device may have contacted hazardous materials, your organization should have a clear release rule. The safe approach is to define categories in advance rather than debating them room by room on pickup day.
How early should we start planning a decommission
Earlier than many realize. Once a shutdown date is fixed, delays usually come from ownership questions, missing asset records, decontamination gaps, and access logistics. Starting early gives you time to separate reusable assets from material recovery and destruction candidates.
What’s the most common reason these programs go off track
Poor internal alignment. Not bad intentions. One team assumes IT cleared the drives, IT assumes the lab removed samples, the lab assumes facilities scheduled access, and the vendor arrives to find unresolved holds. The best projects are the ones where the inventory, release criteria, and paperwork are locked before any equipment moves.
If you need a practical partner for secure, compliant lab and e-waste disposition in metro Atlanta, Scientific Equipment Disposal helps hospitals, universities, research facilities, and government organizations remove and recycle laboratory equipment with on-site logistics, data sanitization, and documented handling that stands up to operational and compliance review.